|
The pooled analysis' findings demonstrated that both primary and secondary indicators had improved. 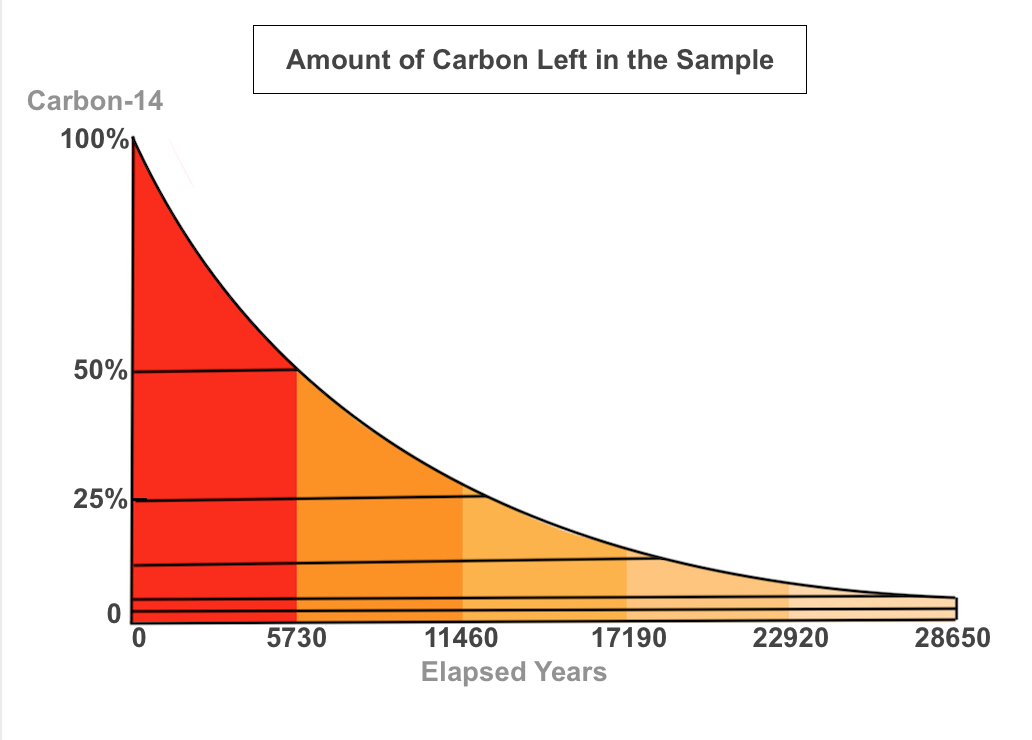
ResultsĪ total of 11 clinical trials met the selection criteria. Statistical analysis was performed using Review Manager 5.4. Outcome indicators of the assessment included liver functions and adverse events. Finally, the data from each study's outcome indicators were extracted for a combined analysis. Literature was screened by the PICOS principle, followed by literature quality evaluation to assess the risk of bias. The data source included PubMed/Medline, Web of Science, EMBASE, and Cochrane Library, from inception to May 2023. 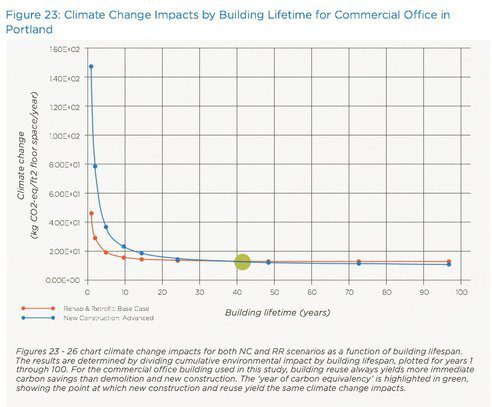
This article aimed to systematically investigate the efficacy and safety of mesenchymal stem cells in the treatment of liver cirrhosis. Clinical cases of mesenchymal stem cell therapy for patients with liver cirrhosis are limited and these studies lack the consistency of treatment effects. Although the efficacy and safety of mesenchymal stem cell therapy for liver cirrhosis have been demonstrated in several studies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
